产品搜索
结构搜索
全站搜索
当前位置: 行业资讯
0D‐in‐1D豌豆荚状CoP@C助力高性能锂离子电池
2018-06-26
来源:转载自第三方
近日,来自北京大学的李星国教授和郑捷副教授课题组在国际期刊Angew. Chem. Int. Ed. 上发表了题为“Peapod‐like CoP@C nanostructure from phosphorization in low‐temperature molten salt for high‐performance lithium ion batteries”的文章[1],利用低毒的等化学计量比的PCl3作为磷源,并首次设计使用了一种低温熔融盐的磷化方法,合成出豆荚状碳包覆CoP纳米材料(CoP@C),用作锂离子电池材料,展现出了巨大的优势。
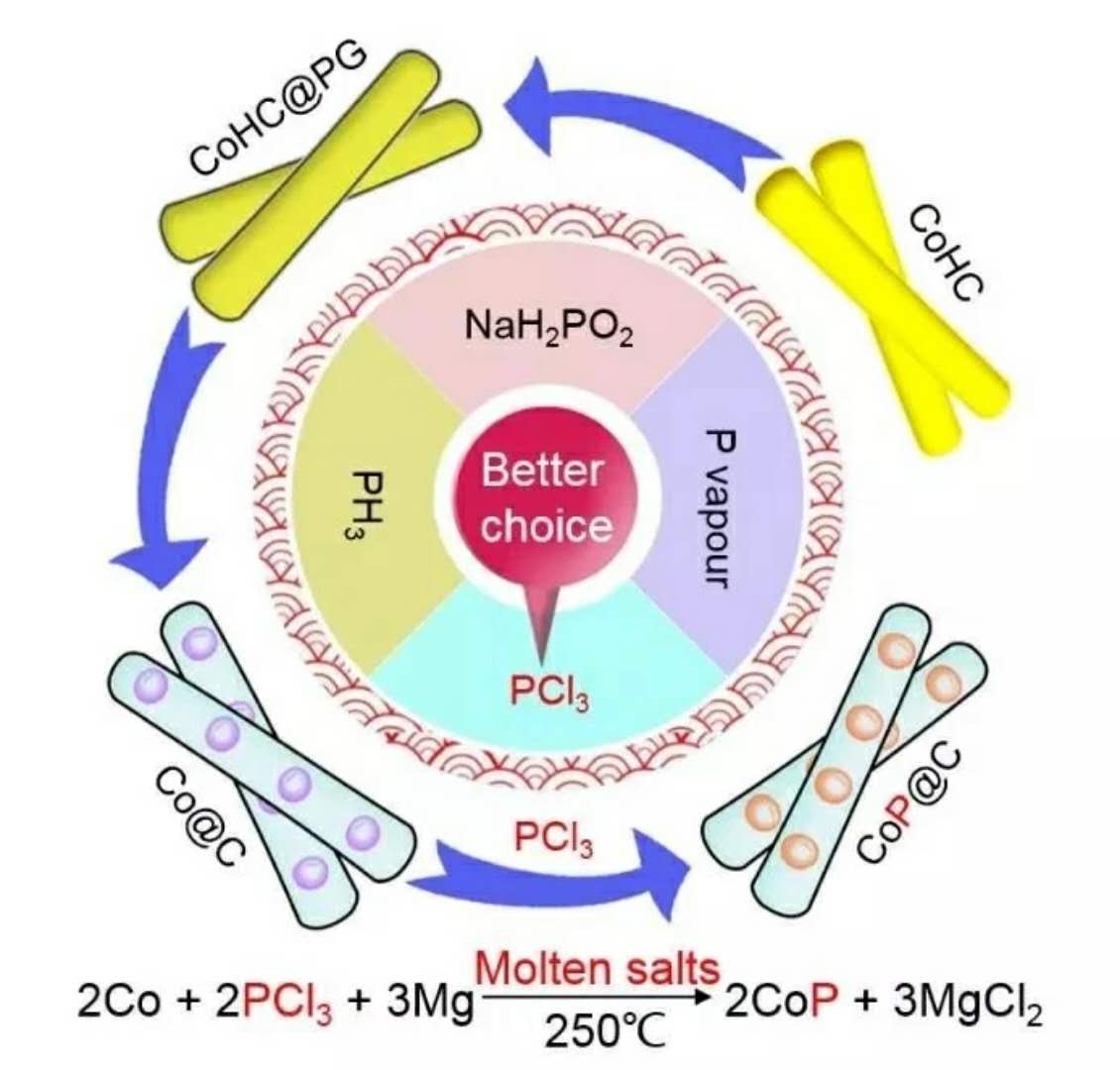
过渡金属磷化物由于其优良的物化性质、低廉的价格以及化学稳定性而受到人们广泛的研究。自20世纪60年代以来,越来越多的科研工作者开始研究过渡金属磷化物作为能量储存和转换的电极材料。目前大多数过渡金属磷化物的制备方法有以下几种:
(1) P蒸汽:由于化学惰性,其磷化通常需要500°C以上高温;
(2) PH3:需要温度低,但是有剧毒,会自燃;
(3) NaH2PO2作为替代的磷源前驱体,加热到350°C可以分解出PH3。
上述磷化方法过程复杂,能耗大,污染严重,并不适于大规模的生产与利用。因此,研究与发展高效制备过渡金属磷化物的方法是储能以及能量转换领域的一个重要研究方向。
在本项研究中,研究人员利用PCl3作为磷源,超低熔点(90°C)的NaCl-KCl-AlCl3熔融盐作为反应介质,Mg作为还原剂,在250°C反应24h,获得了豆荚状CoP@C产物。因为PCl3仅用来完成反应,所以利用更高效;NaCl-KCl-AlCl3熔融盐不仅提供了液体环境,同时通过Al3+/Al氧化还原电对起到良好的电子传递作用,有利于更加安全和高效的无机合成。所获得的CoP纳米颗粒的直径小于50nm,被均匀得包裹在一维的碳纳米管中(CoP@C),伴随着周期性的间隔,是一种良好的一维包覆零维(0D‐in‐1D)的豆荚状结构。
CoP@C结构和物相同时高度可逆转化:CoP@C在嵌入Li+的过程中会逐渐变为1D结构,而脱出Li+之后,又能够变回豆荚状的形态;CoP@C充放电过程中具有相变可逆性,并存在部分锂化的LixCoP过渡相。将其用作锂离子电池负极材料时,锂电池表现出了良好的倍率性能和突出的循环稳定性。
参考文献
[1] Zhiliang Liu, Sungjin Yang, Bingxue Sun, et al. Peapod‐likeCoP@C nanostructure from phosphorization in low‐temperature molten salt forhigh‐performance lithium ion batteries, Angew. Chem. Int. Ed., 2018, DOI: 10.1002/anie.201805468.
本文由苏州亚科科技股份有限公司编辑
如果涉及转载授权,请联系我们。












