产品搜索
结构搜索
全站搜索
当前位置: 行业资讯
CAS:160081-62-9| CDP-Star在诊断试剂盒中的应用
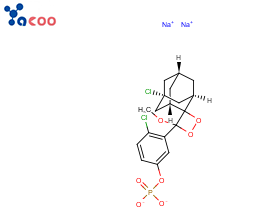
产品名称:2-氯-5-(4-甲氧基螺1,2-二氧杂环丁烷-3,2'-(5-氯三环3.3.1.13.7癸烷)-4-基 -1-苯基磷酸二钠
英文名称:[4-chloro-3-[(3S,5R)-1-chloro-3'-methoxyspiro[adamantane-4,4'-dioxetane]-3'-yl]phenyl] phosphate
英文简称:CDP-Star
CAS:160081-62-9
分子式:C18H19Cl2Na2O7P
货号:C0020
结构式:
产品简介
CDP-Star 是一种碱性磷酸酶(ALP)化学发光底物,由于具有迅速和高灵敏度等特点,通常被用于标记有ALP的生物大分子。CDP-Star 能够通过产生极快的可见光,实现对生物分子的快速、超灵敏检测。近年来,CDP—Star化学发光底物已经被广泛应用于Northern-blot、Southern-blot、Western-blot等印迹技术和免疫分析。
CDP-Star应用领域
艾滋病(AIDS)是由人类免疫缺陷病毒(HIV)感染所引起的一种免疫缺陷性疾病。该病目前尚无有效的治疗方法和预防疫苗,HIV感染诊断进而识别出早期感染的人群是AIDS预防控制工作的首要任务。因此,建立敏感实用的检测方法对于检测、诊断或血液筛查,控制艾滋病的流行就显得非常重要。
目前检测HIV的方法有100多种,最常用的检测HIV抗体的方法有酶联免疫试验(ELISA)、明胶颗粒凝集试验(PA)、免疫荧光分析(IFA)和斑点印迹等。其中ELISA是目前临床上通用的初筛检测方法。但由于其灵敏度低从而受到了限制,后来又发展了灵敏度较高的化学发光免疫分析方法。然而,现有技术中的化学发光免疫分析试剂盒均为封闭式全自动化学发光测量系统,需要昂贵的全自动化学发光测量仪,从而限制了推广使用,无法有效广泛地应用于临床诊断和科研工作。因此,CN101178404A专利提供了一种人类免疫缺陷病毒抗体化学发光免疫分析诊断试剂盒及其制备方法,该试剂盒包括以下组分:
1)抗-FITC抗体包被的固相载体:微孔板、塑料珠、塑料管或磁性颗粒;
2)FITC标记的HIV重组抗原;
3)酶标记的HIV抗原;
4)酶所作用的化学发光底物:(金刚烷)-1,2-二氧乙烷、3-(2′-螺旋金刚烷)-4-甲氧基-4-(3″-磷酰氧基)苯基-1,2-二氧乙烷、CSPD或CDP-Star、鲁米诺或异鲁米诺。
5)阴性对照液:正常人血清。
6)阳性对照液:新生牛血清稀释的阳性混合浆。
与已有的化学发光免疫分析检测方法相比,该方法可以提高分析的灵敏度,均一性和重现性,减少HIV重组抗原的使用量。这种检测方法能在抗体产生初期即检出极微量的病毒抗体,比传统的酶联法检测可提前数天诊断。
参考文献
CN101178404A 人类免疫缺陷病毒抗体化学发光免疫分析诊断试剂盒及其制备方法












