产品搜索
结构搜索
全站搜索
当前位置: 专题聚焦
什么是过渡金属,为什么过渡金属能作催化剂?
2019-06-11
来源:亚科官网
总所周知,燃料电池的核心是将燃料转化为能量的催化剂。催化剂越好,燃料电池的效率越高,功率越高,成本效益越高。过渡金属氧化物是一类具有很大潜力的高性能催化剂,过渡金属催化剂因其来源丰富,价格低廉、催化性能潜力巨大,已经成为当今的研究热点。
什么是过渡金属
过渡金属元素是指元素周期表中d区与ds区(d区元素包括周期系第ⅢB~ⅦB,ⅦI族的元素。不包括镧系和锕系元素。ds区包括周期表第ⅠB~ⅡB族元素。)的一系列金属元素,又称过渡金属。一般来说,这一区域包括3到12一共十个族的元素,但不包括f区(周期表中58~71号元素叫做4f内过渡元素,90~103号元素叫做5f内过渡元素,它们都属于f区元素。)的内过渡元素。
过渡金属由于具有未充满的价层d轨道,基于十八电子规则,性质与其他元素有明显差别。
由于这一区很多元素的电子构型中都有不少单电子(锰这一族尤为突出,d5构型),较容易失去,所以这些金属都有可变价态,有的(如铁)还有多种稳定存在的金属离子。过渡金属最高可以显+7(锰)、+8(锇)氧化态,前者由于单电子的存在,后者由于能级太高,价电子结合的较为松散。高氧化态存在于金属的酸根或酰基中(如:VO43-钒酸根,VO22+钒酰基)。
过渡金属作催化剂的原因
因为过度金属有d轨道电子,或者有空的d轨道,在化学反应中可以提供空轨道充当亲电试剂,或者提供孤对电子充当亲核试剂,形成中间产物,降低反应活化能,促进反应进行。
过渡金属催化剂的特点
1)过渡金属氧化物中的金属阳离子的d电子层容易失去电子或夺取电子,具有较强的氧化还原性能。
2)过渡金属氧化物具有半导体性质。
3)过渡金属氧化物中金属离子的内层价轨道与外来轨道可以发生劈裂。
4)过渡金属氧化物与过渡金属都可作为氧化还原反应催化剂,而前者由于其耐热性、抗毒性强,而且具有光敏、热敏、杂质敏感性,更有利于催化剂性能调变,因此应用更加广泛。
亚科明星产品
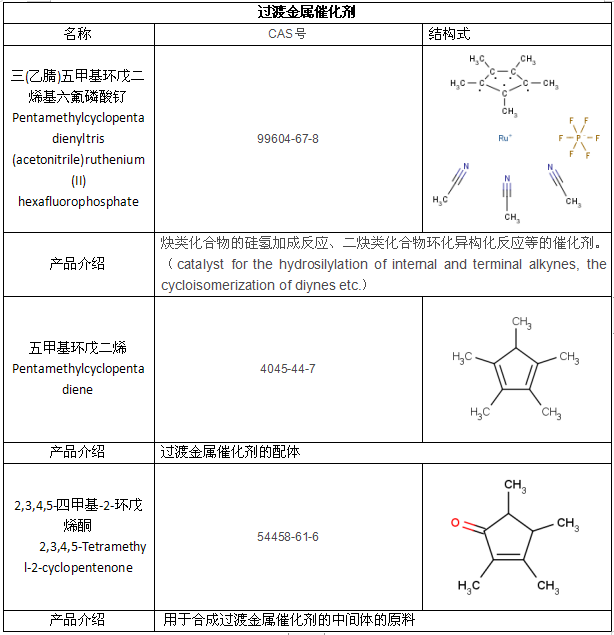
相关链接:过渡金属催化剂
本文由苏州亚科科技股份有限公司编辑












