产品搜索
结构搜索
全站搜索
当前位置: 行业资讯
欧盟批准GLP-1类糖尿病药物Victoza扩展标签
2017-08-01
来源:转载自第三方
2017-8-1
近日,欧盟委员会更新了诺和诺德的糖尿病药物Victoza(liraglutide)的标签。该药物也是欧洲唯一被证明能够改善II型糖尿病血糖水平,同时还可以预防心血管危险的糖尿病药物。
糖尿病是一种慢性病,当胰腺产生不了足够的胰岛素或者人体无法有效地利用所产生的胰岛素时,就会出现糖尿病。如果不加控制,随着时间的推移会对人体的许多系统带来严重损害,如心脏、血管、眼睛、肾脏和神经。糖尿病成人出现心脏病发作和中风的危险会上升2-3倍。
GLP-1是一种由小肠细胞分泌的激素,在进食后能促进胰岛素分泌,抑制胰高血糖素分泌,加快葡萄糖代谢,延缓胃排空,抑制食欲。GLP-1类似物可以有效帮助患者控制血糖水平,同时通过抑制食欲控制体重,达到一箭双雕的效果。与此类似的还有DPP-4抑制剂,其降糖机制与GLP-1类似物对体内血糖的调节机制类似——通过使DPP-4失活而抑制GLP-1被降解,从而发挥降血糖作用。
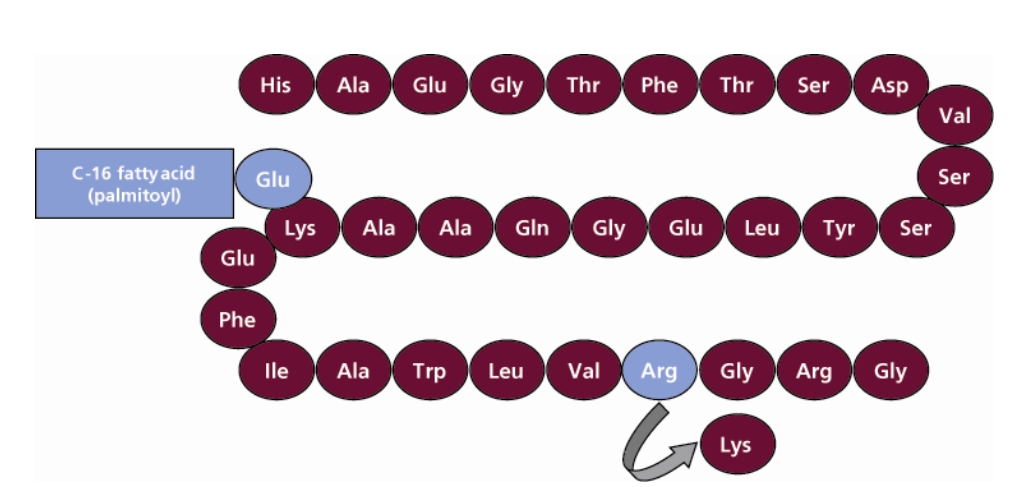
Victoza(liraglutide)是诺和诺德年销20亿美元的糖尿病药物,该药是一种胰高血糖素样肽1(GLP-1)受体激动剂。其在天然GLP-1的分子结构上更换了一个氨基酸,并增加了一个16碳棕榈酰脂肪酸侧链,从而在保留天然GLP-1功效的同时克服了其易降解的缺点。通过这种改变,该药物仅需每日1次注射就能起到良好的降糖作用,并提供多种降糖以外的益处。Victoza于2009年在欧盟批准上市,2010年1月获FDA批准上市,目前已在85个国家上市销售。
去年六月,诺和诺德在美国新奥尔良举行的第76届美国糖尿病协会(ADA)科学会议上公布了Victoza一项大型长期研究LEADER的积极顶线数据。数据显示,与安慰剂相比,Victoza使复合心血管事件(包括心血管死亡,非致死性心肌梗死,非致死性脑卒中)风险显著降低13%,达到了研究的主要终点。该研究中,Victoza的安全性与此前的研究一致。最常见不良事件为胃肠道事件,与安慰剂组相比,Victoza治疗组胰腺炎发病率未明显降低。
糖尿病会增加患者患心脏病、中风等疾病的风险,而心血管并发症反过来也会显著影响糖尿病患者的身体健康及预期寿命。因此,糖尿病治疗药物一直都被寄予希望能降低患者出现心血管并发症的风险。欧盟委员会批准扩大Victoza的药物标签,将提高医生为患者提供GLP-1类药物的治疗机会。
本文由苏州亚科科技股份有限公司编辑
如果涉及转载授权,请联系我们。












